Bacitracin Methylene Disalicylate Soluble Powder 50% is the water-soluble bacitracin methylene disalicylate, with suitable soluble diluent. Bacitracin Methylene Disalicylate Soluble Powder 50% contains 500 g (activity) of bacitracin methylene disalicylate per 1 kg of product for convenient administration through drinking water.
Indications for Use
•Preventionand controlof necrotic enteritis (NE) caused by bacteria susceptible to bacitracin
•Reduction of early mortality in chicks and piglets
•Treatment of swine dysentery associated with microorganisms susceptible to bacitracin
•Control of transmissible enteritis complicated by organisms susceptible to bacitracin
•Reduction of heat stressdue to less heat production
Additional Benefits
•Least influences to beneficial microflora while target pathogens (Clostridium perfringens) remain sensitive to bacitracin in over 50 years
•Improvement of Feed Conversion Rate (FCR) and Growth promotion
• Increase of egg production, eggshell strength, and egg weight
• No residue in drinking water due to an excellent solubility
Recommended Dosage:
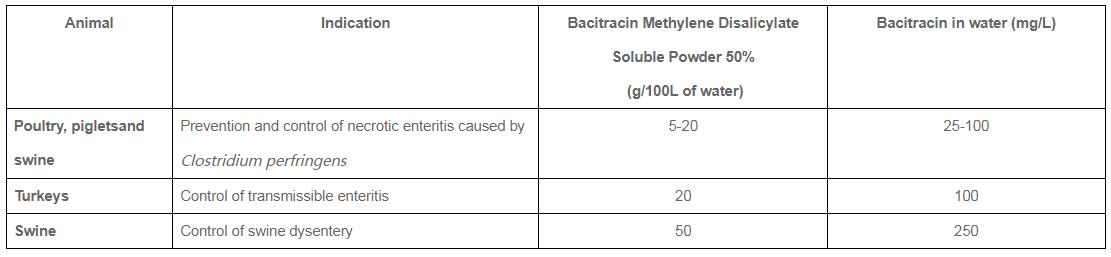
Instruction of Administration
•To prevent an outbreak of disease, start to feed the medicated solution (50-100mg of bacitracin activity/L) at first clinical signs of disease.
•To control necrotic enteritis and transmissible enteritis, administer the medicated solution (100mg of bacitracin activity/L) continuously as long as clinical signs persist.
•To control swine dysentery, start to feed the medicated solution (250 mg of bacitracin activity/L) at first clinical signs of disease continuously until signs of dysentery disappear. Consult a veterinarian if the period of treatment time exceeds 14 days.
Note:The medicated water should be provided continuously as the sole source of drinking water,and should be prepared daily.
Human and Animal Safety
•No toxicity found at 20 times the highest approved level
•No withdrawal period needed
•No cross resistance with other drugs, e.g.,marcrolides, cephalosporins
•No clear evidence of occurrence of antibiotic resistance
•No absorption by animals
Environmental Issues
•Less NH3 and methane emission
•No big impact on environment due to ashort half-life of 2-4 days
•No Salmonella shedding
Compatibility with Other Drugs
•Compatible with all coccidiostats and many other drugs (amprolium, salinomycin, roxarsone, lasalocid, monensin, narasin, ethylpatate, clopidol, decoquinate, nicarbazin, robenidine hydrochloride, zoalene, arsanilic acid, diclazuril, halofuginone, aviax)
•Approved synergic effect with chlortetracycline (CTC)
Other Information
•Self-life is 24 months from the date of manufacture
•Protect contents from humidity after opening, store in a dry and cool place
